
Recent climate predictions suggest East Africa may be in line for an epidemic of Rift Valley fever (RVF) – an infectious disease which can hit people, their livestock and livelihoods, and national economies hard.
Data from the Climate Prediction Centre and the International Research Institute for Climate and Society suggest there is a 90% chance that El Niño will occur towards the end of 2015 to early 2016, and an 85% chance that it will last to early spring 2016. At least two of the most recent RVF epidemics that occurred in East Africa – those in 1997/98 and 2006/2007 – were associated with El Niño weather patterns, with Kenya suffering losses amounting to US$32 million.[1]
El Niño – the warm phase of El Niño Southern Oscillation (ENSO) – is an anomalous warming of sea surface temperatures in central and eastern equatorial Pacific Ocean which occurs cyclically every three to seven years. It follows a weakening of trade winds that usually blow from the east to the west along the equator. The warming of the sea surface temperatures trigger a chain of atmospheric changes throughout the world, resulting in heavy downpours in equatorial Pacific including parts of South America, wet winters in the northern hemisphere and droughts in Indonesia, Philippines and Australia.
The Horn of Africa would experience heavy rainfall particularly when there is a concomitant increase in the sea surface temperatures in the western Indian Ocean. The current predictions give a strong indication that sea surface temperatures in the Indian Ocean are also on an upward trend.
Given the strong predictions of an El Niño occurrence, and the established association between El Niño and Rift Valley fever risk, countries in the Horn of Africa need to start laying out measures to manage the developing risk now. In particular, public education on the linkages between the expected weather patterns and disease risk is vital to minimise human exposure to the disease should an epidemic occur.
Disease and climate change
Questions are always raised on whether there are linkages between ENSO and global warming. ENSO has a short-lived effect on the global temperatures and therefore it is not considered as one of the drivers of climate change, but the strongest naturally occurring source of climate variability globally. Inversely, global warming is likely to double the frequency of ‘super’ El Niños.[2].
Epidemics of malaria, Rift Valley fever and foot rot in animals were reported in eastern Africa during the past El Niño events. Whereas health risks associated with El Niño are episodic (in line with anomalous rainfall), those associated with global warming are steadily increasing. The World Health Organization predicts that the climate-change-induced excess risk of various health outcomes will more than double by 2030.[3]
Conditions for RVF epidemics
RVF is a zoonotic disease (i.e. one that is passed from animals to humans) caused by the RVF virus (RVFV) which is carried by mosquitoes. The intense and persistent rain provides perfect breeding conditions for the mosquito vectors which initiate and intensify RVFV transmission.
Research indicates that floods need to be sustained for at least four to six weeks to allow mosquito populations to reach the critical densities that can initiate an epidemic. Areas that are prone to flooding, such as low-lying regions with flat terrain, as well as those with poor draining soils, are particularly prone to RVF epidemics.[4] Outside the El Niño periods, RVFV occurs in subclinical, inapparent frequencies in the wet lands or riverine vegetation where mosquitoes can still find sites/water to use for breeding. Such areas also tend to have a high diversity of hosts which maintain this (sylvatic) transmission cycle.
RVF epidemics occur if high densities of susceptible hosts are present in an area to permit effective vector-host-virus interactions. Cattle, sheep and goats play a critical role in the amplification of the virus transmission especially during initial stages of the epidemic. Hosts that recover acquire lifetime immunity and would be refractory to RVFV infection in subsequent epidemics.
The higher the proportion of immune animals there are in a population, the less the chances of an epidemic occurring. However, high rates of livestock population turn-over preclude the sustenance of the herd immunity. This could be one of the factors that make areas with higher livestock population turn-over rates, such as arid and semi-arid areas, have more apparent epidemics than other areas.
RVF impacts
RVF is a disease of sheep, goats, cattle and camels characterised by sudden onset of concurrent abortion in multiple species of animals and high mortality rates in the young animals. People get exposed either by coming into direct contact with tissues of infected animals or via mosquito bites. In people, a majority of cases present as acute but mild febrile illness; a few of the cases (less than 8%) result in severe illnesses characterised by haemorrhagic fever, retinitis or encephalitis.[5]
The disease has substantial impacts on livestock productivity and trade. At the farm level, high morbidity and mortality rates result in extensive productivity and food security losses. The entire livestock industry incurs additional losses from market closures, reduced demand for livestock products and embargoes on livestock exports.
It also takes a long time for livestock production to recover following an epidemic because the extensive abortions and perinatal mortality associated with the disease decimate the replacement stock of animals. The implementation of emergency interventions such as surveillance and vaccination campaigns also result in heavy financial losses for countries that are already among the world’s poorest.
Public education and surveillance
Fortunately, several RVF risk management tools have been developed since the last epidemic, such as Contingency Plans, Standard Operating Procedures and Decision Support Framework (DSF), an updated version of which can be found at: https://cgspace.cgiar.org/handle/10568/59781
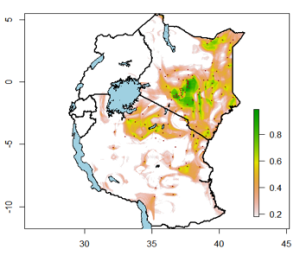
Surveillance, starting with the known RVF hotspots but extending to other areas with time, is also essential. Risk maps can be used to design risk-based surveillance measures have been developed.[6] [7] The one outlined in Figure 1 shows RVF risk spots across East Africa, with areas that were affected in the last epidemic in 2006/2007 overlaid.
Vaccination strategies
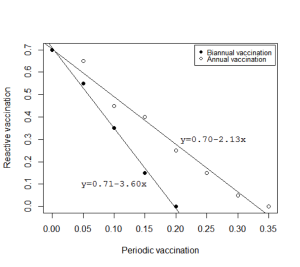
Livestock vaccination should also now be implemented, especially in the hot spots. Currently there are no solid recommendations on how this can be done, but recent model-based analyses implemented as part of Dynamic Drivers of Disease in African Consortium project activities suggest that in areas where there has been no prior or periodic vaccination, a coverage of at least 70% needs to be implemented at least two months before an epidemic (or when the areas are still accessible) to prevent an epidemic occurring. This level of coverage, though, is quite high and may not be feasible for most production systems, especially where livestock producers have a low perception of the risk they face.
These model-based analyses also demonstrate that vaccinating strategically over a five-year period could reduce the thresholds that a reactive vaccination needs to attain to stop an epidemic. Such periodic vaccinations could be done annually or biannually, depending on logistical/economic considerations and the type of vaccine available. This recommendation may not be applicable at the moment when all efforts need to be geared towards managing an impending epidemic, but this approach should be considered in the future.
What is clear now is that there is an urgent need to make the most of existing climate predictions to manage the current risk and some actions such as mass vaccination of livestock have been initiated in various areas. However, in the past, the deployment of response measures has been hampered by the slow processes required to mobilise the required resources. This should not be allowed to happen again. Emergency measures for RVF should be addressed as those of any other adversities that countries should confront.
Ebola and lessons from other recent epidemics
Lessons that have been learned from the recent epidemics of Ebola, bird flu and MERS should be used to inform response to this disease. The recent Ebola epidemic in West Africa for example, demonstrated that a delay in response to such an epidemic would result in a severe health crisis and extensive socio-economic impacts. A few countries such as Nigeria were able to manage the epidemic by responding swiftly and isolating the initial cases. Given the high frequency at which emerging infectious diseases are now occurring, there is a need for early warning systems, preparedness and response plans that identify ways of involving the local communities to effectively manage these risks.
Dr Bernard Bett is a veterinary epidemiologist at the International Livestock Research Institute (ILRI), where he co-leads research on emerging zoonotic diseases in developing countries. He is a partner in the STEPS-led . He works with various actors in the public and private sectors to develop decision support tools for managing disease risks based on information generated from research.
-
Read a STEPS Centre briefing, ‘Rift Valley fever In Kenya: policies to prepare and respond’
-
Find out more about the work of the Dynamic Drivers of Disease in Africa Consortium
Footnotes
[1] Rich, K.M., Wanyoike, F., 2010. An assessment of the regional and national socio-economic impacts of the 2007 Rift Valley fever epidemic in Kenya. Am. J. Trop. Med. Hyg. 83, 52–7. doi:10.4269/ajtmh.2010.09-0291
[2] Cai, W., Borlace, S., Lengaigne, M., van Rensch, P., Collins, M., Vecchi, G., Timmermann, A., Santoso, A., McPhaden, M.J., Wu, L., England, M.H., Wang, G., Guilyardi, E., Jin, F.-F., 2014. Increasing frequency of extreme El Niño events due to greenhouse warming. Nat. Clim. Chang. 5, 1–6. doi:10.1038/nclimate2100
[3] Patz, J. a, Campbell-Lendrum, D., Holloway, T., Foley, J. a, 2005. Impact of regional climate change on human health. Nature 438, 310–317. doi:10.1038/nature04188
[4] Hightower, A., Kinkade, C., Nguku, P.M., Anyangu, A., Mutonga, D., Omolo, J., Njenga, M.K., Feikin, D.R., Schnabel, D., Ombok, M., Breiman, R.F., 2012. Relationship of Climate, Geography, and Geology to the Incidence of Rift Valley Fever in Kenya during the 2006-2007 Epidemic. Am. J. Trop. Med. Hyg. 86, 373–380. doi:10.4269/ajtmh.2012.11-0450
[5] Pepin, M., Bouloy, M., Bird, B.H., Kemp, A., Paweska, J., 2010. Rift Valley fever virus ( Bunyaviridae: Phlebovirus ): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Vet. Res. 41, 61. doi:10.1051/vetres/2010033
[6] Britch, S.C., Binepal, Y.S., Ruder, M.G., Kariithi, H.M., Linthicum, K.J., Anyamba, A., Small, J.L., Tucker, C.J., Ateya, L.O., Oriko, A. a., Gacheru, S., Wilson, W.C., 2013. Rift Valley Fever Risk Map Model and Seroprevalence in Selected Wild Ungulates and Camels from Kenya. PLoS One 8. doi:10.1371/journal.pone.0066626
[7] Anyamba, A., Chretien, J.-P., Small, J., Tucker, C.J., Formenty, P.B., Richardson, J.H., Britch, S.C., Schnabel, D.C., Erickson, R.L., Linthicum, K.J., 2009. Prediction of a Rift Valley fever epidemic. Proc. Natl. Acad. Sci. U. S. A. 106, 955–9. doi:10.1073/pnas.0806490106
[8] Rich, K.M., Wanyoike, F., 2010. An assessment of the regional and national socio-economic impacts of the 2007 Rift Valley fever epidemic in Kenya. Am. J. Trop. Med. Hyg. 83, 52–7. doi:10.4269/ajtmh.2010.09-0291
[9] Cai, W., Borlace, S., Lengaigne, M., van Rensch, P., Collins, M., Vecchi, G., Timmermann, A., Santoso, A., McPhaden, M.J., Wu, L., England, M.H., Wang, G., Guilyardi, E., Jin, F.-F., 2014. Increasing frequency of extreme El Niño events due to greenhouse warming. Nat. Clim. Chang. 5, 1–6. doi:10.1038/nclimate2100
[10] Patz, J. a, Campbell-Lendrum, D., Holloway, T., Foley, J. a, 2005. Impact of regional climate change on human health. Nature 438, 310–317. doi:10.1038/nature04188
[11] Hightower, A., Kinkade, C., Nguku, P.M., Anyangu, A., Mutonga, D., Omolo, J., Njenga, M.K., Feikin, D.R., Schnabel, D., Ombok, M., Breiman, R.F., 2012. Relationship of Climate, Geography, and Geology to the Incidence of Rift Valley Fever in Kenya during the 2006-2007 Epidemic. Am. J. Trop. Med. Hyg. 86, 373–380. doi:10.4269/ajtmh.2012.11-0450
[12] Pepin, M., Bouloy, M., Bird, B.H., Kemp, A., Paweska, J., 2010. Rift Valley fever virus ( Bunyaviridae: Phlebovirus ): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Vet. Res. 41, 61. doi:10.1051/vetres/2010033
[13] Britch, S.C., Binepal, Y.S., Ruder, M.G., Kariithi, H.M., Linthicum, K.J., Anyamba, A., Small, J.L., Tucker, C.J., Ateya, L.O., Oriko, A. a., Gacheru, S., Wilson, W.C., 2013. Rift Valley Fever Risk Map Model and Seroprevalence in Selected Wild Ungulates and Camels from Kenya. PLoS One 8. doi:10.1371/journal.pone.0066626
[14] Anyamba, A., Chretien, J.-P., Small, J., Tucker, C.J., Formenty, P.B., Richardson, J.H., Britch, S.C., Schnabel, D.C., Erickson, R.L., Linthicum, K.J., 2009. Prediction of a Rift Valley fever epidemic. Proc. Natl. Acad. Sci. U. S. A. 106, 955–9. doi:10.1073/pnas.0806490106