
By Kate Jones and Liam Brierley
It might seem strange that after millennia of human history, outbreaks of new, ’emerging’ diseases that we’ve never seen before still regularly occur around the world, some of which go on to become pandemic. However, this may not be so surprising considering how quickly and how intensively the world is changing – expansion of populations, industries, and travel and trade networks are all thought to play a role.
Many current strategies to deal with emerging diseases are reactive, rather than proactive, i.e. the response is focused on dealing with outbreaks after they happen. But what if we could improve our response by projecting where the next outbreak will be and how it will occur? Or better yet, prevent it altogether?
This is the aim of the PREDICT programme, a consortium of worldwide disease biologists. Working with several PREDICT members from the EcoHealth Alliance, our team at University College London, in collaboration too with the STEPS-led Dynamic Drivers of Disease in Africa Consortium, aimed to quantify how these global changes affect the risk of emerging bat-borne viruses.
Bats and disease
The majority of human diseases are zoonoses, that is, they originate via transmission from animals, and bats are no exception. Many devastating viral outbreaks of the last 15 years are suspected to have their origins in bats, from Nipah virus disease in 1999 and SARS in 2002 to the recent outbreaks of Ebola disease and Middle East respiratory syndrome (MERS) in the last several years.
We started by mapping out the potential distribution of each of the 33 viruses shared between bats and humans, collaborating with the Vonhof group at Western Michigan University who have been collecting together data on all viruses known in bats, and which species they infect.
Next, we collected together spatial data on global changes and the wider environment, as well as human density and agriculture. This also included data on domestic animals as bat viruses can sometimes reach people through our livestock, and bushmeat hunting, as bats are hunted and consumed in many parts of the world. It’s critical to understand not just whether these factors affect disease emergence, but also through what means.
Virus risk hotspots
We grouped factors together depending on whether they were likely to increase risk through either increasing richness of viruses (the total number of viruses present) or increasing transmission potential from contact between bats and humans.
Using spatial statistical modelling, we then combined all of this into a single risk map. What we see from this is that, overall, there is a large hotspot of risk in sub-Saharan Africa, including West Africa, where the most recent Ebola virus outbreaks have occurred.
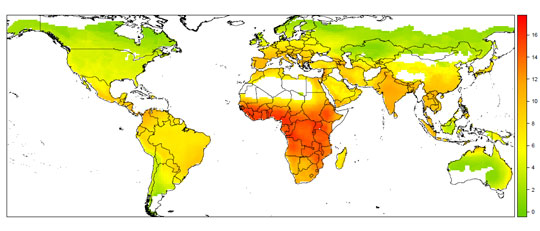
However, when we break down the model into whether the risks occur through richness of viruses or bat-human contact, the resulting maps look very different. Central and South America seem to be a risk hotspot because a naturally high diversity of viruses occurs there, and associations in our model suggest this is a result of a high diversity of bat hosts. Contrastingly, South and East Asia seem to a be a risk hotspot because of high bat-human contact potential, a reflection of high densities of humans and domestic animals, as well as bushmeat practices in some areas.
This tells us something critical – that those places where wild bat populations host many viruses do not seem to be the same as those places where people frequently come into contact with bats, and that both contribute to risk in a different way. Our risk maps take that first step in untangling the complex, multi-step process behind the emergence of a new bat-borne virus in humans.
One Health
In the last decade, there has been a real shift in the way public health is viewed to a much more modern, holistic – or ‘One Health‘ – approach. This acknowledges that animal health and human health are fundamentally connected through our wider ecology, and has guided much of our thinking in the Dynamic Drivers of Disease in Africa Consortium.
Future work will be able to delve deeper into the specific connections between bats and people that could help prevent future disease outbreaks. Although there is much we have yet to understand, what is clear is that bats should not be vilified for their association with emerging diseases. They can be key contributors to environmental stability and ecosystem services, and – admittedly, more sentimentally – they’re fascinating animals, whose world we’ve only just ventured into.
Kate Jones is Professor of Ecology and Biodiversity at University College London and a partner in the STEPS-led Dynamic Drivers of Disease in Africa Consortium. She will be speaking at the One Health for the Real World: zoonoses, ecosystems and wellbeing symposium, being held at the Zoological Society of London, 17-18 March 2016.
‘Quantifying Global Drivers of Zoonotic Bat Viruses: A Process-Based Perspective’, authored by Liam Brierley, Maarten J. Vonhof , Kevin J. Olival, Peter Daszak and Kate E. Jones, is published in The American Naturalist.
Bats are very misunderstood mammals that don’t hurt humans, In fact, they help humans in many ways. I really LOVE them!